-
PRODUCTS/
SERVICES
MMSCs are allogeneic embryonic stem-cell-derived mesenchymal stem cells that have been differentiated and
purified through our company’ s ENS platform technology.
The cells meet all MSC standards as regulated by the International Society for Cellular Therapy (ISCT),
and have been developed and produced to be used for medical therapy.
- Most stem cell therapies all around the world use mesenchymal stem cells (MSC).
- Most stem cell therapies currently being sold are MSC that have been derived from adult cells: bone marrow, cord blood, adipose tissue, etc.
- Mesenchymal stem cells have excellent proliferative capacities, immune system regulation, and anti-inflammatory effects.
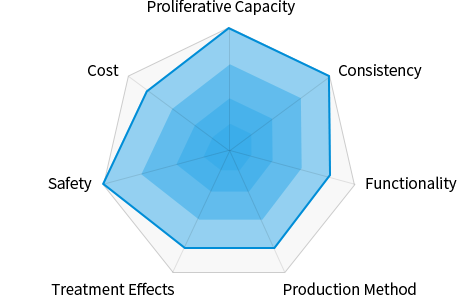
- Proliferative Capacity
- Infinite proliferative capacity
- Consistency
- Almost no deviation
- Functionality
- Abundant secretome
- Production Method
- Consistent, simplified
- Treatment Effects
- Clear MoA
- Safety
- No possibility of tumor formation
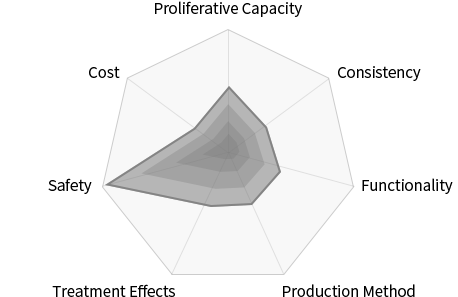
- Proliferative Capacity
- Limited proliferative capacity
- Consistency
- Large deviations dependent on donor
- Functionality
- Limited secretome
- Production Method
- Variable, complicated
- Treatment Effects
- Uncertain MoA
- Safety
- No immune rejection
MMSCs are excellent, high-functional cells that have been shown to be superior to previously used adult stem cells in categories such as proliferative capacity, genetic safety, immunogenicity, anti-inflammatory effects, long-term viability, engraftment, etc. MMSCs also offer the possibility for their indications to be expanded and to be used for various diseases.
-
Urological Disorders
Reliable blood vessel regeneration and tissue regeneration compared to prior adult stem cell therapies
-
Autoimmune Disorders
Strong immune suppression and immune tolerance compared to prior adult stem cell therapies
-
Musculoskeletal Disorders
Conversion to muscle cells, etc.
compared to prior adult stem cell therapies -
Immune Related Disorders
Excellent anti-inflammatory effects compared to prior adult stem cell therapies
-
Cardiovascular Disorders
Long term viability and conversion to muscle cells, etc. compared to prior adult stem cell therapies
-
Attachment

-
Phenotype
-
-
 95%
95%
Positive
(CD73, CD90, CD105) -
-
 2%
2%
Negative
(CD45, CD34, CD19, CD14, HLA-DR)
-
-
-
Differentiation

-
FACS
-
 0.15%
0.15%
-
-
qPCR
- 0.00
-
RNA seq.
- 0
-
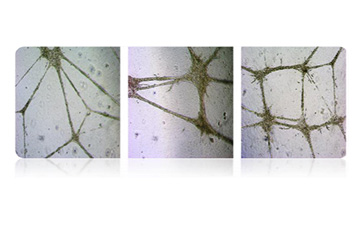
Angiogenesis
-
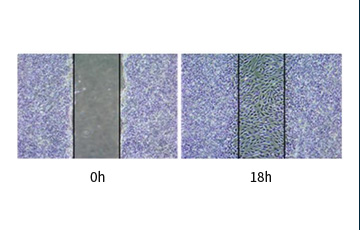
Wound Healing Ability
-
Immune Modulation (BrdU)
-
- 0.8
- MMSC
-
- 0.8
- MMSC + PBMC
-
- 0.9
- PBMC
-
- 1.7
- PBMC + (PBMC + PHA)
-
- 2.2
- PBMC + PHA
-
-
Immune Modulation (IFN-γ)
-
- MMSC
-
- MMSC + PBMC
-
- PBMC
-
- 508.4
- PBMC + (PBMC + PHA)
-
- 1095.5
- PBMC + PHA
-
-
Material Exchange Ability
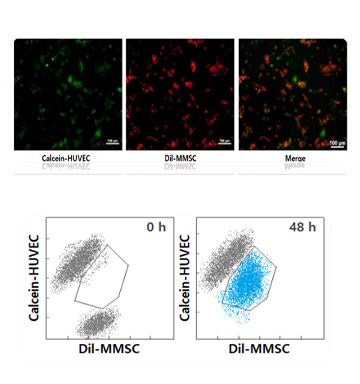
-
- 0h
-
- 46.15%
- 48h
-
-
Wound Healing Ability [Unit: cells]
-
- 90
-
- 330
*migrating cell numbers : about 3.5 times ▲
-
-
Secretion of functional substances
and expression of apoptosis factors [Unit: pg/mL]-
- 1,237
-
- 20,953
* MMP-1 conc. about 17 times ▲
-
- 534
-
- 1,360
* HGF conc. about 2.5 times ▲
-
- 800
-
- 53
* Fas conc. about 15 times ▼
-
-
Proliferative Capacity [Unit: PDL]
-
- 10.17
-
- 16.13
* Cumulative PDL : about 1.6 times ▲
-
-
Cell Size [Unit: μm]
-
- 17.3
-
- 13.6
* about 1.3 times ▼
-
-
Inflammatory and Immune Response
-
- 1
-
* Gata3: about 100.8 times ▲
-
* Adora2a: about 28 times ▲
-
* Gps2: about 11 times ▲
-
* Psma1: about 9.1 times ▲
-
* Pbk: about 7.1 times ▲
-
* Lrfn5: about 7.1 times ▲
-
* Apoe: about 5.5 times ▲
-
* Foxf1: about 4.8 times ▲
-
* Tek: about 4 times ▲
-
* Cx3cl1: about 3.6 times ▲
-
Do you need MMSC samples for the development of medical treatment in the research stage?
Do you need MMSC samples with high secretory abilities to utilize useful substances secreted by stem cells?